Lab 4
Hydrogen Spectrum
Bill Chun Wai Hung
Nimish Kumar
Binh An
Friday Section
7 May 2004
Data:
The experimental results are as the followings.
Table 1. Hydrogen Spectrum
|
Wave Width(nm) |
Wavelength(nm) |
% Intensity |
|
47.320 |
701.975 |
1.00000 |
|
36.736 |
517.754 |
0.09847 |
|
0.760 |
465.071 |
0.06370 |
|
0.357 |
430.430 |
0.05793 |
Table 2. Deuterium Spectrum
|
Wave Width(nm) |
Wavelength (nm) |
% Intensity |
|
44.632 |
700.884 |
1.00000 |
|
38.759 |
519.655 |
0.09951 |
|
1.792 |
463.021 |
0.06052 |
|
1.220 |
431.342 |
0.05843 |
Calculation:
![]() where
where![]()
if the mass of
electron is 9.11 x 10-31 kg,
mass of hydrogen is 1.67 x 10-27 kg, and mass of
deuterium is twice of that of hydrogen, which is 1.67 x 10-27
kg x 2 = 3.34 x10-27.
![]() of hydrogen
of hydrogen ![]()
![]() of deuterium
of deuterium ![]()
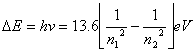
|
Wavelength (nm) |
Relative Intensity |
Transition |
|
383.5384 |
5 |
9->2 |
|
388.9049 |
6 |
8->2 |
|
397.0072 |
8 |
7->2 |
|
410.174 |
15 |
6->2 |
|
434.047 |
30 |
5->2 |
|
286.133 |
80 |
4->2 |
|
656.272 |
120 |
3->2 |
|
656.2852 |
180 |
3->2 |
Conclusion by Bill Chun Wai Hung:
1. Principles and Results
By measuring the radiation emitted from
hydrogen and deuterium, the energy level of hydrogen and deuterium can be
determined. This is because the energy levels of hydrogen and deuterium are
proportional to the intensities of emission of these energy levels.
By using the computer and the light
sensor, graphs of Intensity versus Wavelength of hydrogen and deuterium can be
plotted.
From the graph, information like the
wavelength for certain wavelength, the wave width, and the percentage
intensities can be obtained. The results are presented in Table 1 (Hydrogen)
and Table 2(Deuterium).